NanoSurface Car(ina) Plates,心脏体外测定专用板
型号:内详
联系人:李先生
联系电话:18618101725
品牌:nanosurface?
NanoSurface Car(ina) Plates
Cardiac in vitro assay-ready Plates
Enabling High-throughput Compound Screening with Structurally Matured iPSC-derived Cardiomyocytes
At NanoSurface, we believe that cells in the dish should resemble cells in the body. In the drug development process, the utility of iPSC-based assays is dependent on the physiological relevance and predictive power of the tissue model. Using biomimetic bioengineering techniques to enhance cell-based assays, NanoSurface is working to accelerate the adoption of stem cell technologies early during preclinical drug development, helping get safer medicines to market in less time and at lower cost.
Car(ina) Overview
Step 1: iPSC-derived cardiomyocytes are seeded into assay plates with NanoSurface topography.
Verified compatible with CiPA-validated commercial iPSC-derived cardiomyocyte cell lines of high purity.
Step 2: Cardiac microtissues undergo structural and phenotypic maturation.
Cardiac microtissues undergo structural and phenotypic maturation: tissue alignment and anisotropic cellular development, electrophysiological function, improved sarcomeric development, polarized expression of gap junction proteins (Cx43), and upregulation of protein isoforms (cTnI and β-MyHC).
Step 3: Multiplexed endpoint assays.
NanoSurface Car(ina) Plates are natively compatible with any endpoint assays that use standard microplates. Access metabolic, electrophysiological, contractility, transcriptomic, optical mapping, calcium handling, fluorescence, structural, and biomarker data all in one integrated platform. Compatible with leading commercial MEA instruments.
Key Characteristics

Structurally Matured, Aligned Cardiac Microtissues
Enhanced Reproducibility
High-throughput Screening
Predictive Drug Responses
MEA-based Electrophysiology
Diverse Endpoints
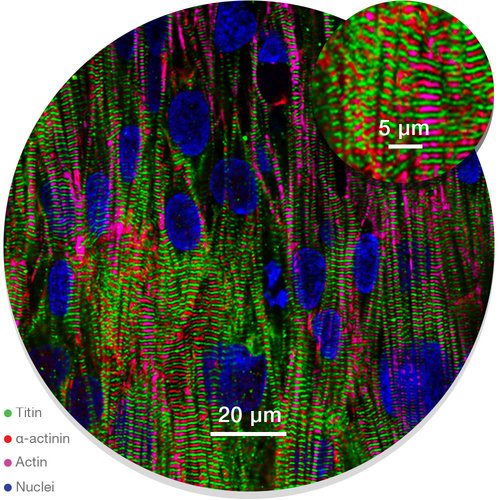
Structurally Matured, Aligned Cardiac Microtissues
NanoSurface Car(ina) tissues comprise iPSC-derived cells that exhibit mature structural and functional phenotypes. Cardiomyocytes cultured on NanoSurface topography exhibit polarized expression of gap junction proteins such as Cx43, develop anisotropic cell shape, striated sarcomeres, tissue-level alignment, as well as achieve enhanced baseline electrophysiology including faster longitudinal conduction velocity and lower resting membrane potential.
Enhanced Reproducibility
Tissue alignment and the development of anisotropic cellular morphology contribute to improvements in well-to-well and experiment-to-experiment reproducibility with decreased variability between cultures.

High-throughput Screening
Car(ina) iPSC-derived cardiac tissues offer improved structural development and enhanced pharmacological utility without compromising high-throughput screening compatibility or ease-of-use. The Car(ina) platform is fully compatible with standard microplate formats and can be directly integrated into existing HTS workflows.
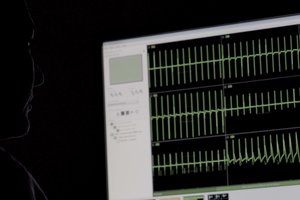
Predictive Drug Responses
Validated to provide predictive responses to drugs of known effect in CiPA-style drug screening applications for the detection of acute cardiotoxicity and pro-arrhythmia. The Car(ina) platform is capable of detecting acute drug cardiotoxicity with enhanced predictive power and sensitivity compared to standard iPSC-based cardiotoxicity assays.

MEA-based Electrophysiology
Structural cues are provided without interfering with electrophysiological signal acquisition during industry-standard MEA and impedance measurements. The Car(ina) platform is fully compatible with high-throughput electrophysiological endpoints.

Diverse Endpoints
Plate-based assays offer uncompromising flexibility in experimental design. The Car(ina) platform is natively compatible with any assay that can be run on a standard microplate. Access metabolic, electrophysiological, transcriptomic, optical mapping, calcium handling, contractility, fluorescence, structural, biomarker, and many more datasets all with one integrated platform.
Toxicity.
Efficacy.
Discovery.

The Car(ina) platform is equipped to improve the physiological relevance of a wide range of applications, such as acute CiPA-style cardiotoxicity screening, chronic cardiotoxicity analysis, structural cardiotoxicity, efficacy screening, drug discovery, and disease modeling.